The HiFAE trial
Home high-flow therapy (HFT) has been shown to reduce acute exacerbation rates and hospital admissions in small studies of patients with COPD.1,2 HiFAE will bring a new level of evidence on the benefits of home HFT in a large cohort of severe COPD patients.
Study objectives
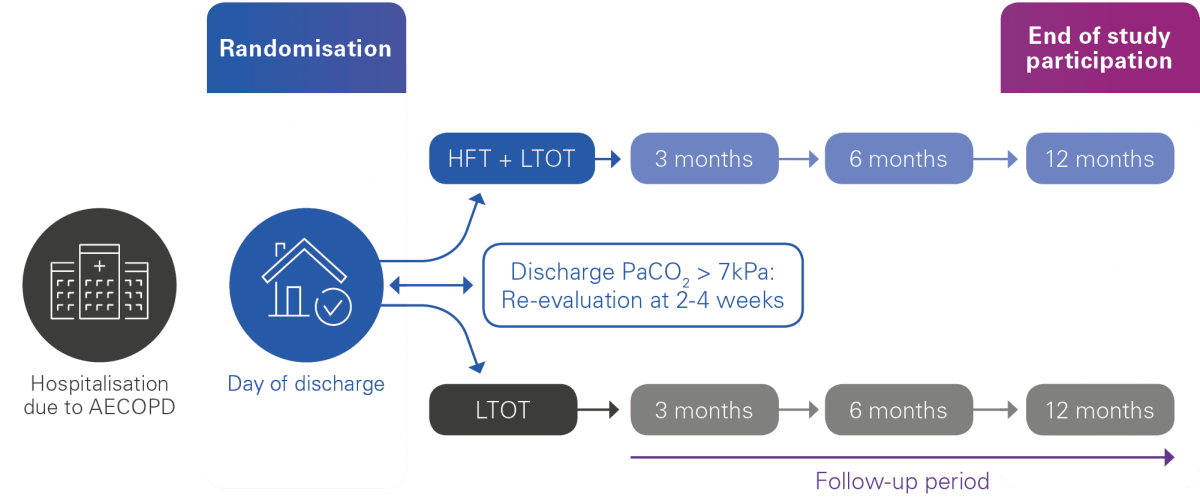
The main objective is to evaluate the efficacy of home HFT with oxygen for reducing severe exacerbations versus standard long-term oxygen therapy (LTOT) in patients previously admitted to hospital for a COPD severe exacerbation. The trial will also evaluate the benefit(s) of home HFT with oxygen on quality of life, respiratory function, exercise capacity, as well as cost effectiveness, compliance and more.
The study endpoints
Primary endpoint:
Time to first hospital admission for AECOPD or death
Secondary endpoints:
The following endpoints will be assessed at 1 year:
- Change from baseline in health-related quality of life
- Change from baseline in gas exchange (PaO2, PaCO2)
- Change from baseline in FEV1 (spirometry)
- Change from baseline in distance walked during the 6-minute walk test
- Clinical course of the disease: all-cause admission-free survival at 1 year, overall survival, exacerbation frequency
- Cost-effectiveness: total costs, cost-utility
- Compliance and tolerance of HFT in the home setting
- Improvement of chronic hypoxemia during follow-up
18 centres


All the news, all in one place
Have you visited our new clinical news page?
Don’t miss out on the latest respiratory research articles and news.
Support for investigator initiated research
Resmed believes in the need to support ethical, independent clinical research, conducted by qualified third-party investigators.
This content is intended for health professionals only
* Please see https://clinicaltrials.gov/ct2/show/NCT05196698 for further information
** Programme Hospitalier de Recherche Clinique (PHRC) du Ministère des solidarités et de la santé. Please see https://solidarites-sante.gouv.fr/systeme-desante-et-medico-social/innovation-et-recherche/l-innovation-et-la-recherche-clinique/appels-a-projets/article/programme-hospitalier-de-recherche-clinique-phrc for further information
- Storgaard LH, et al. Long-term effects of oxygen-enriched high-flow nasal cannula treatment in COPD patients with chronic hypoxemic respiratory failure. Int J Chron Obstruct Pulmon Dis 2018;13:1195-1205. DOI : 10.2147/COPD.S159666
- Rea H, et al. The clinical utility of long-term humidification therapy in chronic airway disease. Respir Med 2010;104:525-533. DOI : 10.1016/j.rmed.2009.12.016