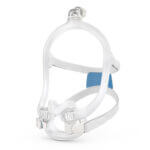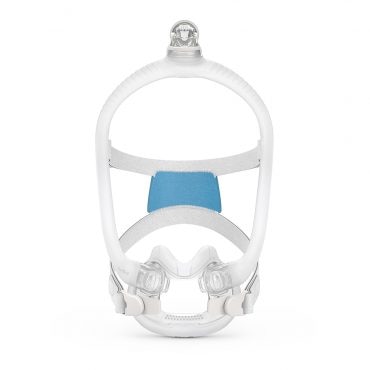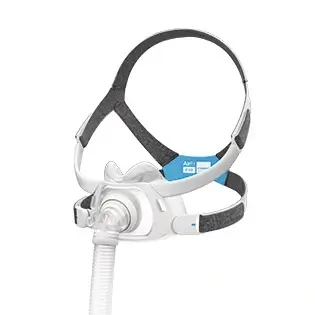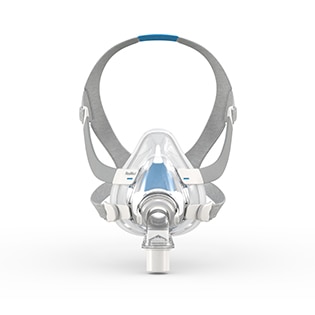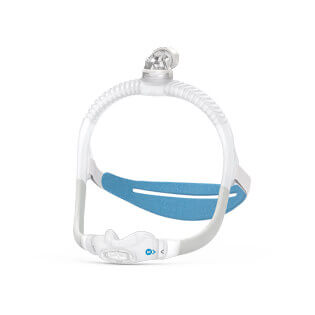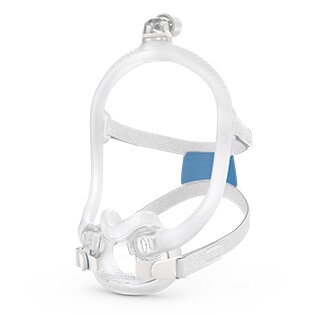
AirFit™ F30i
Full face mask for CPAP therapy
The AirFit F30i is a tube-up full face mask featuring a comfortable under-the-nose cushion. You can sleep in the position you want1 and say goodbye to red marks on your nasal bridge. AirFit F30i is part of the AirFit 30 series range of under-the-nose masks.
The mask contains magnets that may interfere with certain implants or medical devices. Please refer to the user guide for complete information, including magnet contraindications and warning.
A non-magnetic AirFit F30i mask is also available, suitable for people who require a mask without magnets.
Availability may vary across regions
Sleep how you choose
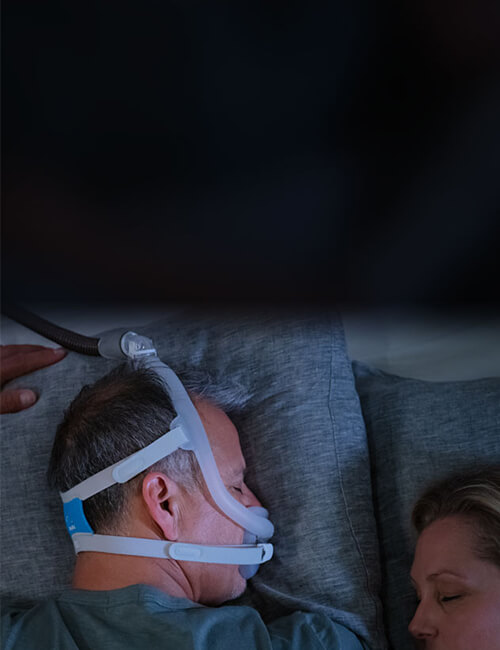
With the AirFit F30i full face mask, it’s easy to sleep in any position you choose.1 When you sleep on your front, you’ll appreciate the clutter-free design with no front-end tube connection.
Easy-to-use features

The AirFit F30i CPAP mask features magnetic clips* and a quick-release elbow to make life easy. Taking the mask on and off is quick1, convenient and intuitive, with magnetic clips that guide the headgear to the frame swiftly.
Nothing over your nose

The F30i’s ultra-compact cushion sits comfortably under your nose, not over it.1 Less facial contact looks good, feels good and means you can say goodbye to red marks and pressure on the bridge of your nose.
How to buy
Please contact our Customer Service department or your local representative for more information on obtaining ResMed products.
How do I fit my AirFit F30i full face mask?
How do I assemble/dissassemble my AirFit F30i mask?
How do I inspect and clean my AirFit F30i mask?
Interested in exploring other mask options?
How do you sleep? What makes you feel more comfortable? Our mask categories help you identify a CPAP mask that matches your sleep needs and preferences.
Please refer to the user guides for relevant information related to any warnings and precautions to be considered before and during use of the products.
*Contraindication
Masks with magnetic components are contraindicated for use by patients where they, or anyone in close physical contact while using the mask, have the following:
- Active medical implants that interact with magnets (i.e., pacemakers, implantable cardioverter defibrillators (ICD), neurostimulators, cerebrospinal fluid (CSF) shunts, insulin/infusion pumps)
- Metallic implants/objects containing ferromagnetic material (i.e., aneurysm clips/flow disruption devices, embolic coils, stents, valves, electrodes, implants to restore hearing or balance with implanted magnets, ocular implants, metallic splinters in the eye)
Warning
Keep the mask magnets at a safe distance of at least 6 inches (150 mm) away from implants or medical devices that may be adversely affected by magnetic interference. This warning applies to you or anyone in close physical contact with your mask. The magnets are in the frame and lower headgear clips, with a magnetic field strength of up to 400mT. When worn, they connect to secure the mask but may inadvertently detach while asleep. Implants/medical devices, including those listed within contraindications, may be adversely affected if they change function under external magnetic fields or contain ferromagnetic materials that attract/repel to magnetic fields (some metallic implants, e.g., contact lenses with metal, dental implants, metallic cranial plates, screws, burr hole covers, and bone substitute devices). Consult your physician and manufacturer of your implant / other medical device for information on the potential adverse effects of magnetic fields.
References:
1. ResMed external clinical study of 51 current full face CPAP mask users, conducted between 24/10/2018 – 20/11/2018, 2018 data on file; ID A4484728.
Content last updated: 10/2023.